The growth of South Africa’s medical device market has been largely driven by increased healthcare demand due to an aging population and a rise in chronic diseases. The country has a well-developed infrastructure and economy, along with a strong healthcare system, especially in urban areas. Government initiatives like the National Health Insurance system and investments in healthcare infrastructure continue to boost demand for medical devices.
Furthermore, South Africa’s strategic location offers easy access to other African markets, while its robust private healthcare sector drives the adoption of new technologies. The country is a hub for research and development and attracts medical tourism, further increasing the need for advanced MedTech.
South Africa has established a robust regulatory framework for medical devices and in vitro diagnostic (IVD) devices, overseen by the South African Health Products Regulatory Authority (SAHPRA). This framework emphasises patient safety, quality assurance, and product efficacy, aligning closely with international standards to facilitate both domestic innovation and global market access.
Below, in part four of our fast-growing markets series, our Head of Regulatory Bruno Gretler shares an overview of South Africa’s regulatory landscape for medical devices and IVDs, covering classification, market access pathways, post-market surveillance, and strategies to overcome the key challenges.
An overview of the South African regulatory framework
SAHPRA, established under the Medicines and Related Substances Act of 1965, is responsible for the regulation of medical devices and IVDs in South Africa, ensuring compliance through pre-market authorisation, licensing, and post-market monitoring. The regulatory approach is risk-based, meaning higher-risk devices undergo rigorous evaluations, while lower-risk products follow streamlined approval pathways. This methodology is consistent with international best practices as guided by the International Medical Device Regulators Forum (IMDRF), the World Health Organisation (WHO), the FDA, and the EU Medical Device Regulation (EU MDR).
Despite the progress achieved, the regulatory landscape faces challenges such as resource limitations and procedural complexities. In response, SAHPRA has initiated digital transformation projects, increased stakeholder collaboration, and actively participates in global regulatory discussions to refine practices and promote transparency and efficiency.
Complementing SAHPRA’s efforts, the Department of Health and the South African Bureau of Standards (SABS) play significant roles in technical standardisation and enforcement. Together, these institutions continuously enhance the regulatory environment, balancing stringent safety standards with the promotion of technological innovation and industry growth.
How are medical devices classified in South Africa?
The classification of medical devices and IVDs in South Africa is essential for defining market access requirements. SAHPRA’s approach ensures regulatory oversight is proportionate to potential risks.
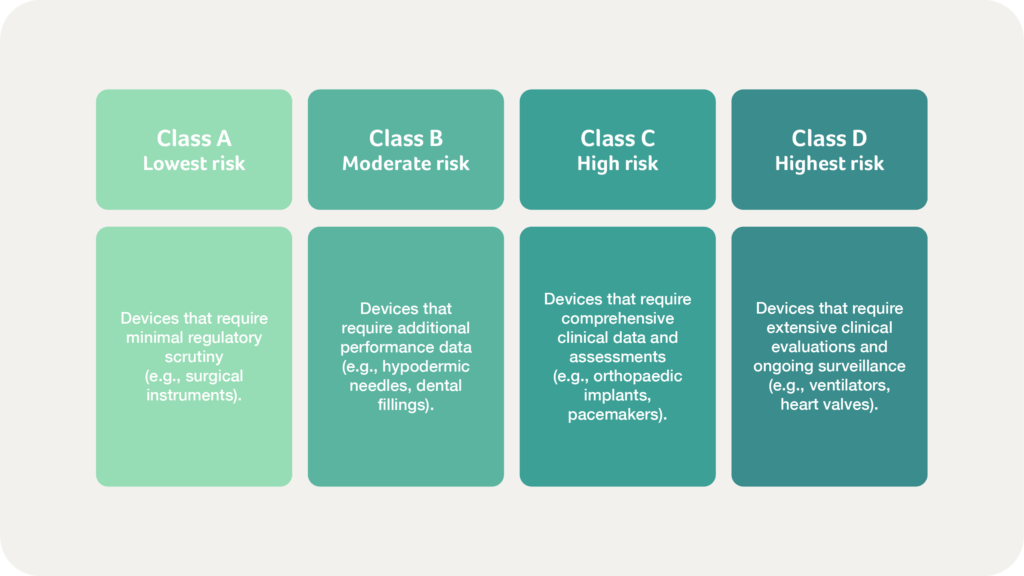
Medical devices are classified into four risk-based categories:
How are IVDs classified in South Africa?
Similarly, IVD devices follow the same risk classification:
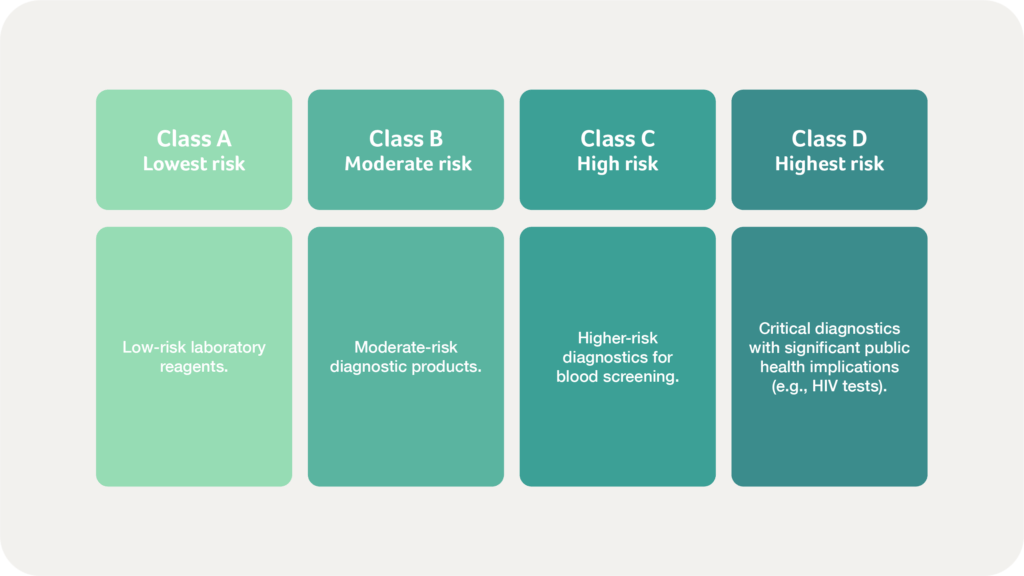
Accurate classification determines regulatory pathways, conformity assessment requirements, and surveillance needs. Manufacturers must ensure correct device classification to streamline market entry and maintain compliance. SAHPRA continuously revises its guidance to reflect new technology and evolving risk profiles, so we recommend regularly referring to their official guidelines (SAHPRA Medical Devices Classification) for the latest updates.
What does the South African market pathway for medical devices & IVDs involve?
The pathway to market for medical devices and IVDs in South Africa is a structured regulatory process designed to ensure compliance with national and international safety and efficacy standards. The process encompasses multiple steps, beginning with classification and licensing and extending through pre-market approval, conformity assessments, and post-market obligations.
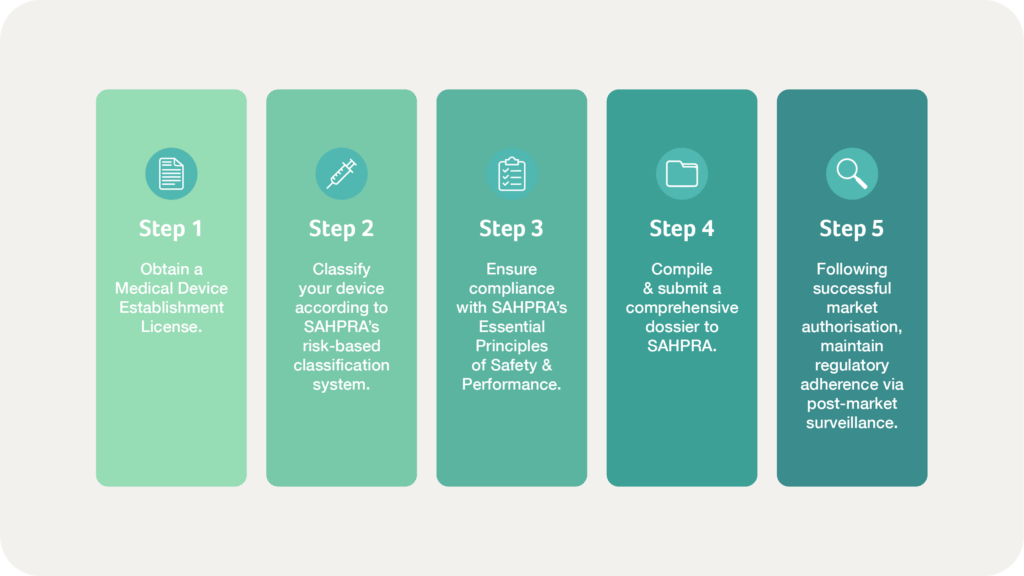
Step 1 | Obtain a Medical Device Establishment License
The first step in bringing a medical device or IVD to market involves obtaining a Medical Device Establishment License. Any entity involved in the manufacture, importation, distribution, or sale of these products must secure this license from SAHPRA, demonstrating adherence to a recognised quality management system such as ISO 13485. This requirement ensures that companies operate within an established regulatory framework that prioritises product safety and performance.
Step 2 | Classify your device
Once an establishment is licensed, manufacturers must classify their products according to SAHPRA’s risk-based classification system, which aligns with global standards. As noted above, classification determines the level of regulatory scrutiny, with Class A devices (lowest risk) requiring minimal oversight and Class D devices (highest risk) undergoing extensive regulatory evaluation. IVDs follow a similar classification process, considering the implications of incorrect test results on patient care and public health.
Step 3 | Comply with Essential Principles of Safety & Performance
Compliance with SAHPRA’s Essential Principles of Safety and Performance is a fundamental requirement for all medical devices and IVDs. These principles align with global regulatory standards and mandate that products must be designed and manufactured to ensure intended performance without compromising patient safety.
Manufacturers can demonstrate compliance through technical documentation, clinical evaluations, risk assessments, and adherence to standards set by the International Electrotechnical Commission (IEC) or International Organisation for Standardisation (ISO).
Step 4 | Compile & submit a comprehensive dossier
The submission and review process requires manufacturers to compile and submit a comprehensive dossier to SAHPRA. This dossier includes detailed information about the device’s intended use, design specifications, manufacturing processes, risk management strategies, and clinical evidence where applicable.
Lower-risk devices may undergo an expedited review, whereas higher-risk devices typically require more rigorous evaluations, including clinical performance data and additional conformity assessments conducted by recognised certification bodies.
Step 5 | Implement continuous post-market surveillance
Upon successful review and approval, SAHPRA grants market authorisation, permitting the legal marketing and sale of the medical device or IVD in South Africa. However, compliance does not end with market entry. Manufacturers and distributors must maintain regulatory adherence through continuous post-market surveillance, timely reporting of adverse events, and updates to SAHPRA regarding any modifications to their products. Any significant changes to a device’s design, labelling, or intended use may necessitate further regulatory review and approval.
What post-market requirements should be considered for the South African market?
Post-market surveillance (PMS) is integral to South Africa’s regulatory oversight of medical devices and IVDs, ensuring ongoing safety, effectiveness, and quality. Manufacturers must actively monitor device performance in real-world settings and promptly report serious adverse events to SAHPRA for swift evaluation and regulatory response, including corrective actions or recalls.
Key PMS responsibilities include:
- Systematic monitoring of adverse events and product complaints
- Prompt reporting of serious incidents to SAHPRA, enabling timely regulatory action
- Submission of Periodic Safety Update Reports (PSURs) for high-risk devices, providing continuous risk-benefit assessments based on real-world data
- Collaboration with SAHPRA in conducting inspections, audits, and investigations to reinforce compliance and maintain public trust
To ensure consistent reporting for adverse events, we recommend implementing effective data management systems and digital reporting tools. Strengthening stakeholder collaboration and digital surveillance capabilities further enhances responsiveness to safety concerns.
Effective PMS ensures early identification of rare or unforeseen issues, reinforces public confidence, and maintains regulatory integrity. It is crucial for the ongoing assessment of device performance and safety, thereby supporting public health and fostering trust in the healthcare system.
What are the challenges unique to the South African regulatory landscape?
Despite the robust, structured nature of South Africa’s medical device regulatory framework, challenges such as lengthy approval times and complex documentation requirements can create barriers to market entry.
Regulatory complexity and frequent policy updates require manufacturers and regulators alike to continually adapt and refine their practices. Alignment with international standards, such as those set by the IMDRF, helps global market access but demands extensive resource investment and specialised knowledge.
Manufacturers often encounter difficulties due to resource constraints, particularly small and medium-sized enterprises (SMEs), which struggle with the price and logistics associated with generating local clinical data. Conducting local clinical trials, often mandatory for regulatory submissions, can be costly and resource intensive.
How to overcome the regulatory challenges in South Africa
Proactively engage with SAHPRA
Strategies to navigate these challenges include proactive engagement with SAHPRA early in the product lifecycle. By ensuring your team is monitoring the landscape for any regulatory updates, your organisation can stay up to date on the latest guidance and policies and strategically factor any changes into your regulatory planning.
Forge strategic partnerships
Relying on strategic partnerships with academic institutions and research organisations for clinical data support can assist with resource challenges.
Think beyond borders
Adherence to international best practices can help mitigate the challenges noted above, facilitating a smoother approval process. Furthermore, participating in multi-regional clinical trials can spread costs and enhance efficiency.
Utilise funding opportunities
Accessing government incentives or industry-led funding programs can further alleviate financial strain for smaller manufacturers.
More broadly speaking, continued industry-regulator collaboration and investments in capacity building, infrastructure development, and regulatory training will ensure sustained innovation, improved compliance, and enhanced market access for medical device manufacturers in South Africa.
In conclusion…
South Africa’s regulatory framework for medical devices and IVDs is continuously advancing, increasingly aligning with international standards while effectively addressing local healthcare requirements. Although challenges like prolonged approval timelines and complex documentation requirements persist, SAHPRA’s proactive efforts in digital transformation, transparency, and global harmonization are creating a more efficient and reliable regulatory environment.
Manufacturers can benefit significantly from early and proactive engagement with SAHPRA, strategic compliance planning, and leveraging tactical partnerships. By adopting these strategies, stakeholders can effectively navigate the regulatory landscape and streamline market access into South Africa.
Are you looking to register your medical device in South Africa? Our Regulatory team is ready to help – simply get in touch to start the conversation.
Our fast-growing markets series…
Our fast-growing markets series also covers regulatory guides for Brazil, Mexico, and India.






