
ISO 10993-1:2025 | What’s new in the revised standard?
Back in June, our Senior Operations Consultant and Biocompatibility Specialist Dr. Silvio Peng shared five important updates from the draft ISO…
Congenius

Back in June, our Senior Operations Consultant and Biocompatibility Specialist Dr. Silvio Peng shared five important updates from the draft ISO…
Congenius

ISO 10993-1 provides the governing foundation for the biological evaluation of medical devices, explaining the fundamental requirements and general principles.…
Congenius

In September 2024, the FDA published its draft guidance on “Chemical Analysis for Biocompatibility Assessment of Medical Devices” to improve…
Congenius

In the realm of medical technology and innovation, the development and production of safe and effective medical devices is of…
Congenius

Whether for economic or strategic reasons, medical device manufacturers often outsource their production to another site or external company. In…
Congenius

The Analytical Evaluation Threshold (AET) is the crucial parameter for demonstrating that your analytical results sufficiently assess the biological safety…
Congenius
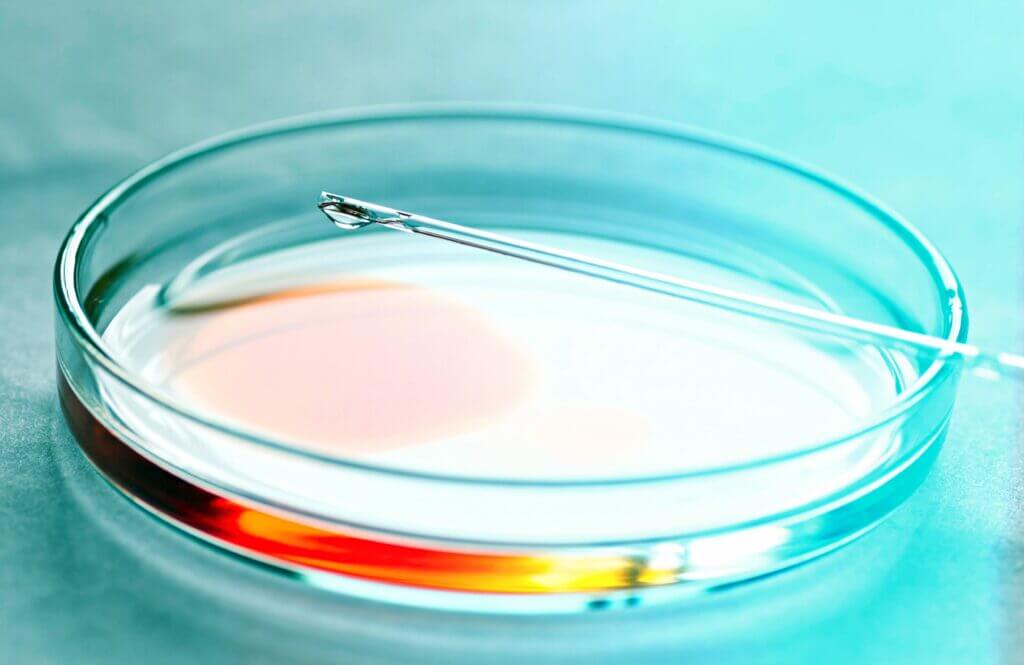
ISO 10993-23:2021 Biological evaluation of medical devices – Part 23: Tests for irritation assesses the possible contact hazards from medical…
Congenius

In 2017, cobalt was up-classified by the European Chemicals Agency (ECHA) with respect to its carcinogenic, reproductive toxicity and mutagenic…
Congenius